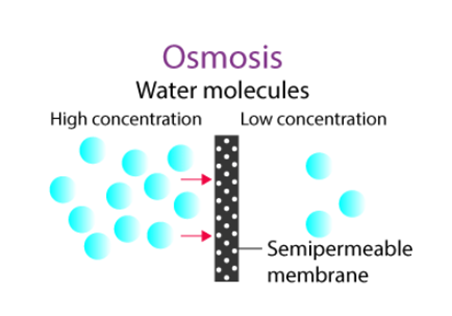
There are two ways to transfer water across a semipermeable membrane: osmosis and reverse osmosis. The semipermeable membrane may be a form of polymeric membrane that is biological or synthetic that permits just specific molecules or ions to diffuse across it.
Osmosis and reverse osmosis differ primarily in that osmosis involves the diffusion of water molecules across a semipermeable membrane from a high to a lower water potential, whereas reverse osmosis involves the diffusion of water molecules across a semipermeable membrane against the potential gradient. Additionally, although reverse osmosis is an artificial process, osmosis is a natural process.
Here we are going to discuss each of the process separately for better understanding of the recent technology used in water treatment processes, let’s get started..
Osmosis:
Osmosis is the diffusion of a solvent through a membrane with variable levels of permeability. Water is often the solvent in biological processes. Every time the water concentrations are different on each side of a differentially permeable membrane, osmosis will happen. Water molecules pass via a potential gradient by osmosis. It happens across a semipermeable membrane, primarily the cell’s plasma membrane. Until the water potential on either side of the semipermeable membrane is equal, water molecules shift from a higher to a lower water potential. Endosmosis and exosmosis are the two primary forms of osmosis that may take place within a cell.
Endosmosis: When cells are put in a hypotonic solution, which has a greater water potential than the cytosol, endosmosis happens. Overfilling with water can occasionally cause cells to break open.
Exosmosis: When cells are put in a hypertonic solution, which has a lower water potential than the cytosol, exosmosis happens.

Osmosis is crucial for maintaining homeostasis between the inside and outside environments. As an illustration, consider the osmosis of water from the earth to the plant’s root hairs. Water travels from the soil to a location of low concentration because the soil contains a higher concentration of water (ie. Plant)
Applications of osmosis:
The stomata opening, which occurs during plant gas exchange
Roots’ ability to draw water from the soil
Effects of introducing freshwater and saltwater fish to water with varying salt concentrations
Reverse osmosis:
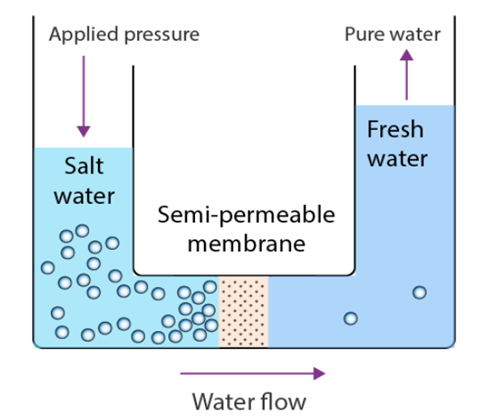
One of the oldest and most well-liked separation methods, reverse osmosis is mostly employed to purify water. The method was primarily used for the desalination of seawater in 1950, when it was still a very slow process and only used in a few labs. However, there have been substantial breakthroughs as a result of extensive study and technological advancements, particularly in the area of polymers and the creation of effective membranes.
Today, a lot of people utilise this method to purify water for commercial, residential, industrial, and scientific uses. Although reverse osmosis technology is a significant scientific advancement of humanity, we shall gain a fundamental grasp of the entire process on this page.
Reverse Osmosis principle:
To further simplify the process, consider the fact that because a membrane is present, big molecules of the solute cannot pass through it and instead remain on the side that is under pressure. On the other hand, the membrane does not prevent the pure solvent from passing through. When this occurs, the solute’s molecules begin to concentrate on one side of the membrane while getting diluted on the other. Additionally, there is considerable variation in the levels of solutions.
Fundamentally, reverse osmosis occurs when the solvent crosses the membrane against a gradient of concentration. Essentially, it shifts from one concentrate to the next.
Reverse Osmosis Benefits:
Here are some of the advantages of reverse osmosis:
1. Many different kinds of dissolved and suspended chemical particles, as well as biological agents (like bacteria), may be successfully removed from water using this method.
2. This method is widely used to handle liquid discharges or pollutants.
3. It helps to avoid sickness by cleaning water.
4. It aids in saltwater desalination.
5. The medical industry benefits from it.
Why choose Netsol!
Membranes with Netsol water solutions improve product yields while preserving consistency and quality Reverse osmosis, nanofiltration, ultrafiltration, and microfiltration are just a few of the technologies that are covered by Netsol water solution’s spiral wrapped membranes, which are made to fulfil the demands of several applications.
As leaders in water and waste water treatment for more than a decade, we offer complete water solutions for businesses, homes, and communities. Planning, integrating, and managing water on the supply, quality, quantity, discharge, and environmental fronts are all part of our end-to-end responsibilities. Process technology, design engineering, and project management skills are used to achieve this. We give our clients observable benefits to demonstrate the importance of efficient water management.
If you are curious to know more about the best RO manufacturing company in your vicinity, feel free to contact us an at +91-9650608473 or enquiry@netsolwater.com.
